Philippines, US trialling repurposed drugs to fight mild Covid-19
In the US, the search is on for an existing prescription or over-the-counter medication that could help treat people with mild to moderate symptoms of Covid-19.
Just In
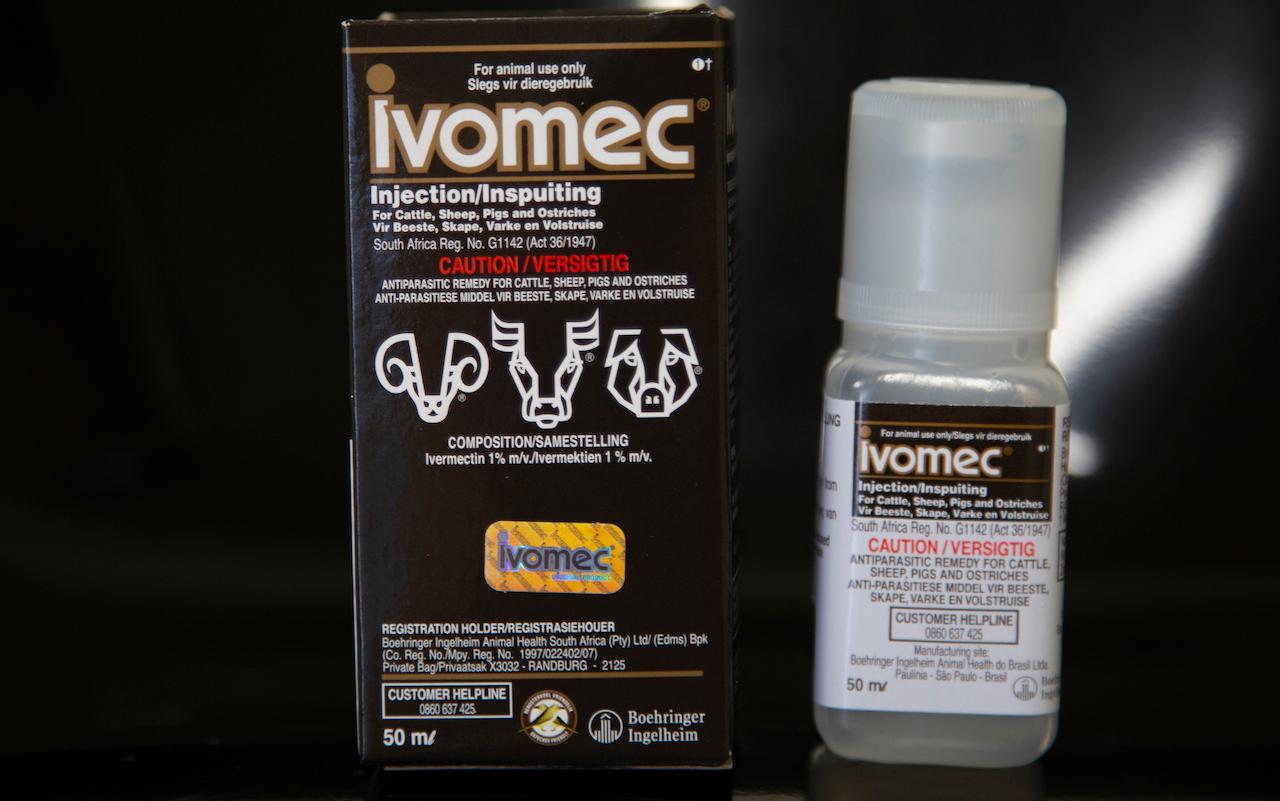
The Philippines will begin clinical trial of several drugs, including the anti-parasite medication ivermectin, in patients with Covid-19 to determine their efficacy in combating the coronavirus, a senior government official said.
The nation is facing one of the worst coronavirus outbreaks in Asia, battling a renewed surge in infections.
Some politicians have started promoting the use of ivermectin for coronavirus and have given out free doses although the country’s food and drugs regulator has cautioned against its use because of a lack of evidence for the drug as a treatment.
The clinical trial of ivermectin, which could last for six months, “will give us a more reliable estimate of its effects as an anti-viral agent in mild and moderate Covid-19 patients”, Science and Technology Minister Fortunato Dela Pena said in a presentation late on Tuesday.
Ivermectin tablets have been approved for treating some worm infestations and for veterinary use in animals for parasites.
The World Health Organization last month recommended against using ivermectin in patients with Covid-19 except for clinical trials, because of a lack of data.
Dela Pena said the government has also approved the clinical trials of a new formulation of methylprednisolone, a steroid, and melatonin, as possible treatments for Covid-19.
The government will also start trials of a herbal supplement, derived from the native tawa-tawa plant that can fight dengue, he said, adding to ongoing tests using virgin coconut oil for severe Covid-19 patients.
“We are trying several medications. They may not be vaccines, but they could potentially speed up the recovery,” Dela Pena said.
In the US, the National Institutes of Health (NIH) announced on Monday funding for a large clinical trial to see if existing prescription and over-the-counter medications could help treat people with mild to moderate symptoms of Covid-19.
NIH director Francis S Collins said he hopes the trial will find a way to allow adults who are not sick enough to be hospitalised with Covid-19 to be able to treat themselves with medications that are already commonly available to the public.
He said, “While we’re doing a good job with treating hospitalised patients with severe Covid-19, we don’t currently have an approved medication that can be self-administered to ease symptoms of people suffering from mild disease at home, and reduce the chance of their needing hospitalisation.”
Subscribe to our newsletter
To be updated with all the latest news and analyses daily.