China approves 2 more home-grown Covid-19 vaccines for use
China is exporting vaccines to 27 countries and providing free doses to 53 others, a foreign ministry spokesman says.
Just In
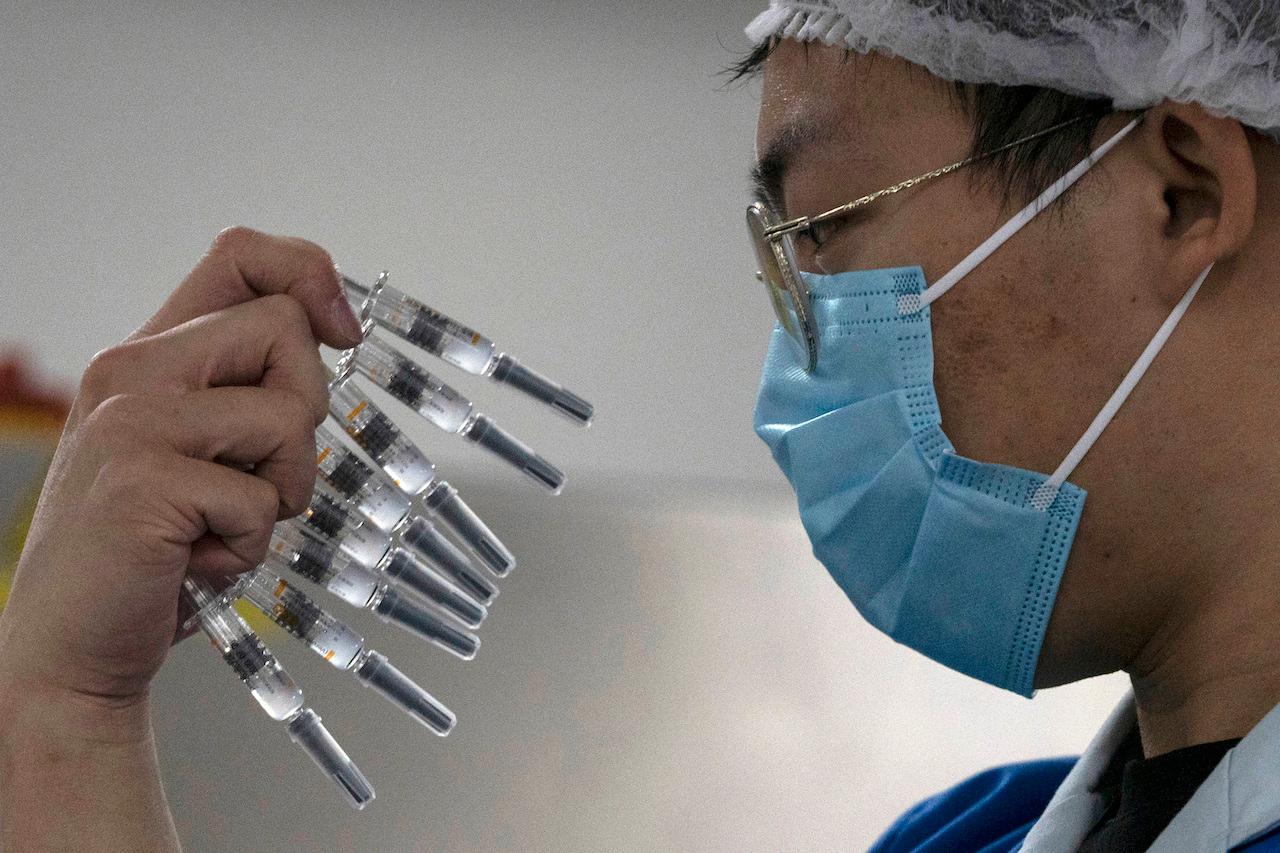
China’s medical products regulator said on Thursday that it has approved two more Covid-19 vaccines for public use, raising the number of domestically produced vaccines that can be used in China to four.
The two newly cleared vaccines are made by CanSino Biologics and Wuhan Institute of Biological Products, an affiliate of China National Pharmaceutical Group (Sinopharm).
They join a vaccine from Sinovac Biotech approved earlier this month, and another from Sinopharm’s Beijing arm approved last year.
Prior to formal approval for wider public use by the National Medical Products Administration, millions of doses of the two Sinopharm vaccines and Sinovac shot were administered in China’s vaccination programme.
The four approved Chinese vaccines can all be stored at normal freezer temperatures – a potential selling point in less affluent countries that might have difficulty deploying vaccines from Pfizer and Moderna that require much colder temperatures for longer-term storage.
The three companies have all secured overseas supply deals, and China has donated doses made by Sinopharm’s Beijing unit to numerous other nations.
Altogether, China is exporting vaccines to 27 countries and providing free doses to 53 others, a foreign ministry spokesman said on Tuesday.
So far, China has not approved any Covid-19 vaccines developed by Western drug makers.
Subscribe to our newsletter
To be updated with all the latest news and analyses daily.